 This gives us an answer of 255.36 joules. Whereas in the adiabatic case, the internal energy had to increase. Substituting these values in, we find that the work done is equal to 28000 pascals times 0.042 cubic meters minus 0.03288 cubic meters. Two mole of ideal gas diatomic gas (C v, m 5 / 2 R) at 3 0 0 K and 5 atm expanded irreversely and adiabatically to a final pressure of 2 atm against a constant pressure of 1 atm. This means the internal energy has to drop. Is this a correct intuition about the adiabatic versus isothermal case?Īs for the adiabatic vs isobaric case, as you compress the gas, in order for the pressure to stay the same, the temperature has to drop. raises temperature), which means you have to do more work still to compress the gas down further.  In an adiabatic process, however, the energy you transfer to the gas by doing work on it (compressing it) has no way of leaving the container, so it stays there and makes the molecules more energetic (i.e. Compression of the gas adds energy to the gas, meaning that energy came from the environment and was put into the gas via work. IfW1is the work done in compressing an ideal gas from a given initial state through a certain volume isothermally andW2is the work done in compressing the same gas from the same initial state through the same volume adiabatically, then: (1)W1W2 (2)W1W2 (4)W12W2 Thermodynamics Physics Practice questions, MCQs, Past Year Questions (PYQs), NCERT Questions, Question Bank, Class 11 and. In an adiabatic process, however, the energy you transfer to the gas by doing work on it (compressing it) has no way of leaving the container, so it stays there and makes the molecules more energetic (i.e. Compression of the gas adds energy to the gas, meaning that energy came from the environment and was put into the gas via work. IfW1is the work done in compressing an ideal gas from a given initial state through a certain volume isothermally andW2is the work done in compressing the same gas from the same initial state through the same volume adiabatically, then: (1)W1W2 (2)W1W2 (4)W12W2 Thermodynamics Physics Practice questions, MCQs, Past Year Questions (PYQs), NCERT Questions, Question Bank, Class 11 and.
It will be 'sapped out' of the gas by a cold bath reservoir or something of that nature. In an isothermal process, this energy is not allowed to increase the gas's temperature. The reason for this is that, as you press on the gas to reduce its volume, you increase its energy by doing work on it. More work will be required in compressing the gas adiabatically than isothermally. I'm trying to reason it out 'physically' rather than 'algebraically' using formulas.Īs I said, I'm trying to reason this through physically, by imagining what happens to the gas molecules and their energy as the volume decreases. I'm deliberately trying to avoid using equations.  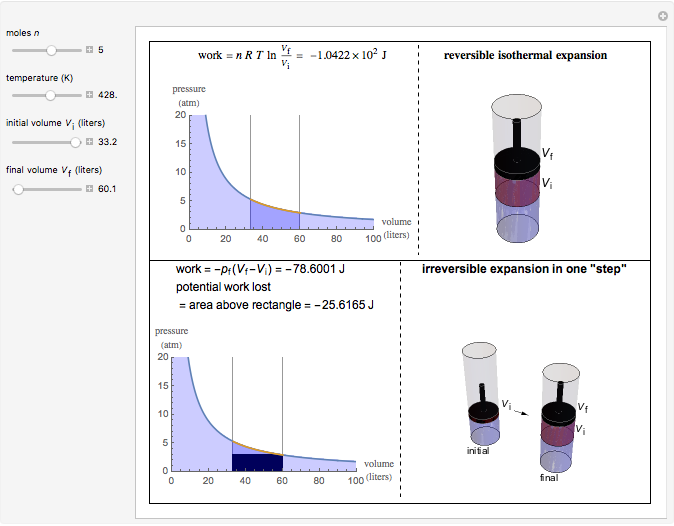 Thermodynamics is a branch of physics which deals with the energy and work of a system. Medium Solution Verified by Toppr Given that the process is reversible adiabatic.  Compute the final volume reached by gas in this case. If W, is the work done in compressing an ideal gas from a given initial state through a certain volume isothermally and W, is the work done in compressing the. Which of the following processes results in the most work done on the gas? a) isothermal b) adiabatic c) isobaric d) The work done is independent of the process. 287 kPa m /kg K)(27 273 K) 3 3 v RT P The gas constant corresponds to the work performed by one mole of an ideal gas when heated by 1 K at constant pressure. For a gas, work equals change in pressure times a change in volume. Calculate workdone in adiabatic compression of one mole of an ideal gas (monoatomic) from an initial pressure of 1 atm to final pressure of 2 atm. calculate the work done by the system when the temperature of the gas is. \[\left( \frac\)).An ideal gas is compressed to half its initial volume by means of several possible processes. Well learn later that for an ideal gas to maintain its temperature while expanding, an amount of heat must be added to the gas that is equal to the work done. According to the ideal gas law, pressure varies linearly with temperature and. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
